CBSE Class 10 Science PYQ 2023
Chemical Reactions and Equations Previous Year Questions
Set 31/1/1 | Important MCQs with Answers
Practice CBSE Class 10 Science Chemical Reactions and Equations PYQ 2023 with MCQs, assertion-reason and detailed answers.
Q1. In the experimental setup given below, it is observed that on passing the gas produced in the reaction in the solution ‘X’ the solution ‘X’ first turns milky and then colourless.
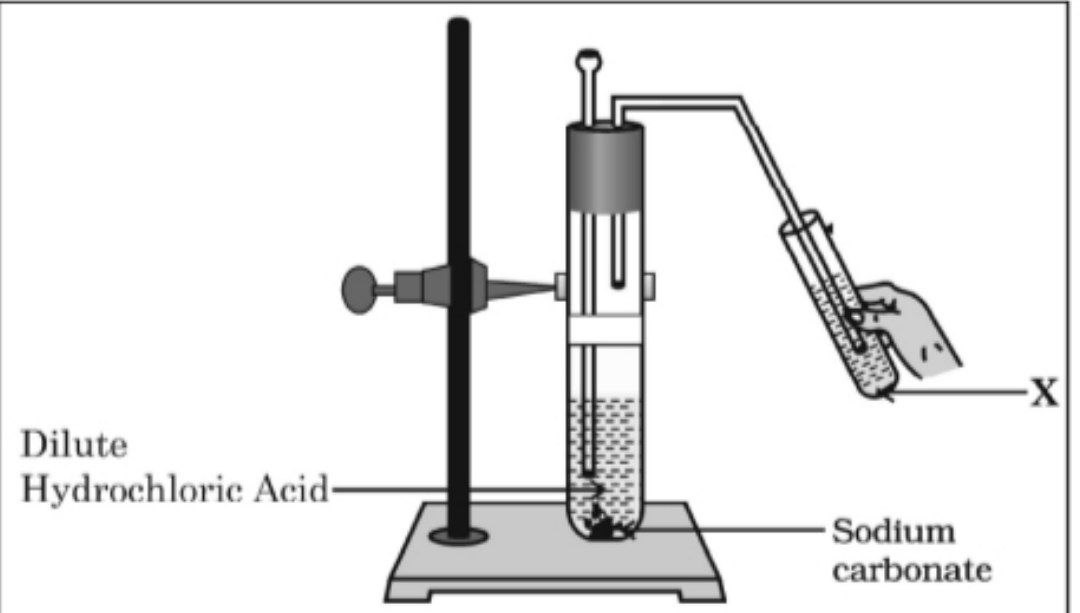
The option that justifies the above stated observation is that ‘X’ is aqueous calcium hydroxide and
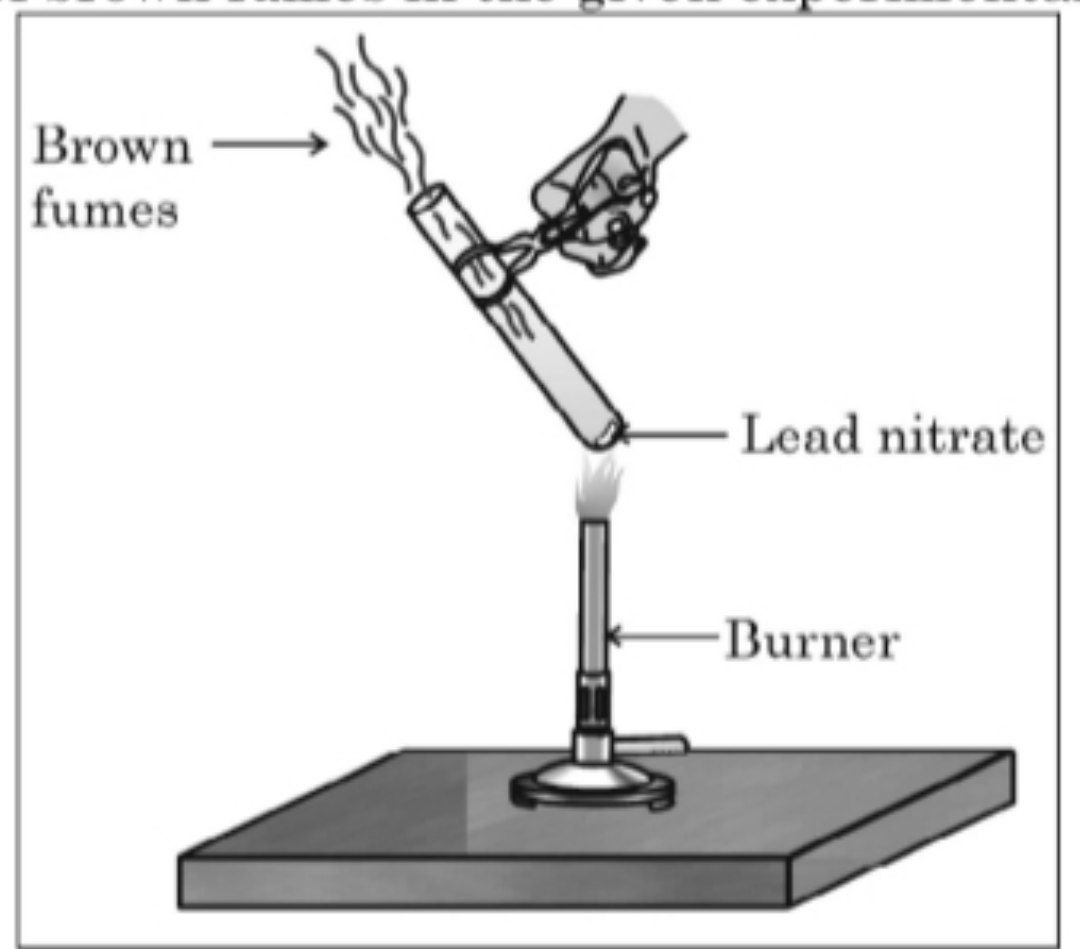
Q2. The emission of brown fumes in the given experimental set-up is due to
Q3. MnO₂ + xHCl → MnCl₂ + yH₂O + zCl₂ In order to balance the above chemical equation, the values of x, y and z respectively are:
Q17. Assertion (A): The colour of aqueous solution of copper sulphate turns colourless when a piece of lead is added to it. Reason (R): Lead is more reactive than copper, and hence displaces copper from its salt solution.
Q27. (a) Identify the reducing agent in the following reactions : (i) 4NH₃ + 5O₂ → 4NO + 6H₂O (ii) H₂O₂ + F₂ → HF + HOF (iii) Fe₂O₃ + 3CO → 2Fe + 3CO₂ (iv) 2H₂ + O₂ → 2H₂O
(b) Define a redox reaction in terms of gain or loss of oxygen.
(a)
(i) NH₃
(ii) H₂O₂
(iii) CO
(iv) H₂
(b) A reaction in which gain or loss of oxygen takes place simultaneously is called a redox reaction.
Q37. Almost all metals combine with oxygen to form metal oxides. Metal oxides are generally basic in nature. But some metal oxides show both basic as well as acidic behaviour. Different metals show different reactivities towards oxygen. Some react vigorously while some do not react at all.
(a) What happens when copper is heated in air? (Give the equation of the reaction involved.)
(b) Why are some metal oxides categorized as amphoteric? Give one example.
(c) Complete the following equations : (i) Na₂O(s) + H₂O(l) → ______ (ii) Al₂O₃ + 2NaOH → ______
(a) 2Cu + O₂ → 2CuO
(b) Amphoteric oxides react with both acids and bases to produce salt and water.
Example: Al₂O₃ / ZnO
(c)
(i) Na₂O + H₂O → 2NaOH
(ii) Al₂O₃ + 2NaOH → 2NaAlO₂ + H₂O
2023 – Code 31/2/2
Q1. During electrolysis of water, if the volumes of oxygen and hydrogen evolved at the electrodes are VO and VH respectively, then VO/VH is
Q5. In order to balance the following chemical equation, the values of the coefficients x and y respectively are :
x Pb(NO₃)₂ → 2PbO + y NO₂ + O₂
Q23. (a) What is observed when aqueous solutions of potassium iodide and lead nitrate are mixed together ? Name the type of reaction and write the chemical equation for the reaction that occurs.
OR
(b) When copper powder is heated in a watch glass, a black substance is formed. (i) Why is this black substance formed ? Name it. (ii) How can this black substance be reversed to its original form ?
(a) Yellow precipitate of lead iodide is formed.
Type: Double displacement / Precipitation reaction
Pb(NO₃)₂ + 2KI → PbI₂ + 2KNO₃
OR
(b)
(i) Oxygen is added to copper / Copper is oxidised.
Substance: Copper oxide (CuO)
(ii) By passing hydrogen gas over it
CuO + H₂ → Cu + H₂O
Q27. Write down the balanced chemical equations for the following reactions and identify the type of reaction in each case.
(a) Nitrogen gas is treated with hydrogen gas to form ammonia gas. (b) Lead nitrate is heated strongly to form lead monoxide, nitrogen dioxide and oxygen. (c) A copper wire is dipped in silver nitrate solution and a shining deposit of silver is produced.
(a) Combination reaction
N₂ + 3H₂ → 2NH₃
(b) Decomposition reaction (Thermal decomposition)
2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂
(c) Displacement reaction
Cu + 2AgNO₃ → Cu(NO₃)₂ + 2Ag
2023 – Code 31/4/3
Q1. When aqueous solutions of potassium iodide and lead nitrate are mixed, an insoluble substance separates out. The chemical equation for the reaction involved is :
✔ Marking Scheme: 1 mark
Q2. When Sodium bicarbonate reacts with dilute hydrochloric acid, the gas evolved is :
✔ Marking Scheme: 1 mark
Q3. Acid present in tomato is :
✔ Marking Scheme: 1 mark
Q18. Assertion (A): Reaction of Quicklime with water is an exothermic reaction. Reason (R): Quicklime reacts vigorously with water releasing a large amount of heat.
✔ Marking Scheme: 1 mark
Q22. (A) A student took a small amount of copper oxide in a conical flask and added dilute hydrochloric acid to it with constant stirring. He observed a change in colour of the solution.
(i) Write the name of the compound formed and its colour. (ii) Write a balanced chemical equation for the reaction involved.
OR (B) The industrial process used for the manufacture of caustic soda involves electrolysis of aqueous solution of compound ‘X’. In this process, two gases ‘Y’ and ‘Z’ are liberated.
(A)
(i) Copper(II) chloride (CuCl₂), blue-green colour
(ii) CuO + 2HCl → CuCl₂ + H₂O
✔ Marking Scheme: ½ + ½ + 1 = 2 marks
OR
(B)
X = Sodium chloride (NaCl)
Y = Hydrogen (H₂)
Z = Chlorine (Cl₂)
B = Bleaching powder (CaOCl₂)
✔ Marking Scheme: ½ + ½ + ½ + ½ = 2 marks
2023 – Code 31/5/3
Q1. The balanced chemical equation showing reaction between quicklime and water is :
✔ Marking Scheme: 1 mark
Q4. Study the following chemical reaction : 2Na (s) + 2H₂O (l) → 2NaOH (aq) + H₂ (g) ↑ The reducing agent in this reaction is :
✔ Marking Scheme: 1 mark
Q18. Assertion (A): In the following reaction ZnO + C → Zn + CO ZnO undergoes reduction. Reason (R): Carbon is a reducing agent that reduces ZnO to Zn.
✔ Marking Scheme: 1 mark
Q27. (a) Write a balanced equation to show the reaction that occurs when a piece of aluminium is dipped in : (i) Dilute sulphuric acid (ii) Sodium hydroxide
(b) Write the colour of the solution formed when copper oxide is treated with hydrochloric acid. Give reason for this observation.
(a)
(i) 2Al + 3H₂SO₄ → Al₂(SO₄)₃ + 3H₂ ↑
(ii) 2Al + 2NaOH → 2NaAlO₂ + H₂ ↑
✔ Marks: 1 + 1
(b)
Colour: Blue-green
Reason: Due to formation of Copper(II) chloride (CuCl₂)
✔ Marks: ½ + ½
Total = 3 marks
Q28. (a) Define a double displacement reaction. (b) Write the chemical equation of a double displacement reaction which is also a (i) Neutralization reaction (ii) Precipitation reaction. Give justification.
(a) A reaction in which there is an exchange of ions between the reactants.
✔ Marks: 1
(b)
(i) HCl + NaOH → NaCl + H₂O
Neutralization: Acid reacts with base to form salt and water
✔ Marks: ½ + ½
(ii) Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl
Precipitation: Insoluble BaSO₄ is formed
✔ Marks: ½ + ½
Total = 3 marks
